
The electronic structures of atoms help explain the properties of elements and the structure of the periodic table.
HELIUM ELECTRON CONFIGURATION FULL
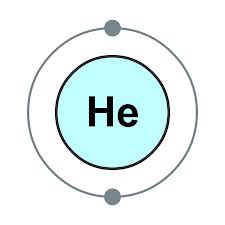
These elements have full outer shells - helium is 2, neon is 2.8, argon is 2.8.8, and their group number is group 0Įlectronic structures and properties of elements Helium, neon and other elements in this group Has only 1 electron but not placed in group 1 as it does not share properties with the rest of the group - it is placed by itself on the periodic table Note that there are some exceptions to the pattern shown with the example above.
How do I write an electron configuration? See the examples below for a detailed explanation.Ģ. How do I determine the electron configuration?Ĭheck out the periodic table to find out how many electrons an atom has, draw out the progression of subshells, and fill the orbitals of the energy levels one by one until all electrons have a place. An element’s group number on the periodic table is equivalent to the number of electrons in its outermost shell.ġ.

The different shells can hold different numbers of electrons. Refresher: Electrons exist in shells, orbiting around an element’s nucleus.
